D-Z-No-Phi

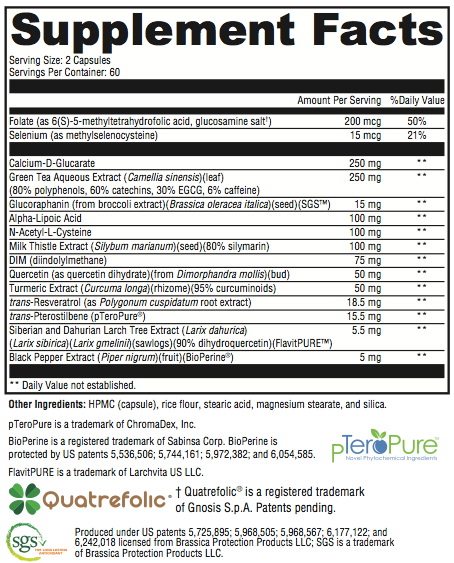
Antioxidant and Detoxification Support Several nutrients support antioxidant activity, both phases of detoxification, and the health and function of the liver (the major site of detoxification). Milk thistle extract contains silymarin, a compound found to limit the entry of hepatotoxins, donate sulfhydryl groups for detoxification, and increase hepatic glutathione by over 35%.[7] Its action in the liver reduces fat peroxidation and fibrous tissue formation, supports a normal immune and inflammatory response, promotes protein synthesis and tissue regeneration, and supports glucuronidation and glutathione levels.[8] Alpha-lipoic acid is both water- and fat-soluble. It supports glutathione metabolism, helps regenerate antioxidant vitamins C and E, and helps maintain the ratio of reduced-to-oxidized CoQ10 in mitochondria.[7] The redox couple of lipoic acid and dihydrolipoic acid stabilizes NF-kappaB transcription and may help support healthy immune functions in the body.[9,10] Methylselenocysteine (MSC) is considered a well-tolerated form of the trace element selenium and may support normal cell-life regulation.[11] Selenium provides antioxidant support via glutathione peroxidase and manganese superoxide dismutase (MnSOD) activity.[12] N-acetyl-cysteine (NAC) may significantly increase glutathione in the body, which, in turn, is incorporated into crucial antioxidant and detoxification enzymes. Glutathione supports antioxidant activity, phase II detoxification, and the normal breakdown of metabolites, toxins, and other compounds. NAC supports phase II sulfation reactions as well.[7] Calcium D-glucarate has been added to support glucuronidation. 5-methyltetrahydrofolate (5-MTHF) is present as Quatrefolic® (a stable, bioavailable form of folate) to support methylation, energy generation, and phase l and phase ll activity.*
Phytonutrients A variety of phytonutrients support antioxidant activity in the body. Green tea catechins have been found to assist in free-radical scavenging, support detoxification through modification of phase I and phase II enzymes, and support normal cell-life regulation via multiple signaling pathways.[13,14] Bioflavonoids, including resveratrol, quercetin, and the highly absorbable FlavitPURETM form of dihydroquercetin (DHQ), support phase I detoxification as well as intermediary antioxidant protection.[1] Pterostilbene, a highly absorbable, methylated form of resveratrol, is thought to work together with quercetin in supporting normal cell-life regulation.[15] Turmeric extract provides curcumin, a phytonutrient valued for its promotion of antioxidant activity, support of metabolic detoxification, and modulation of cytokine production.[16,17] BioPerine®, a patented form of piperine from black pepper, has been added to enhance the absorption of nutrients, particularly curcumin.*[18]
Xenoestrogen Metabolism D-Z-No-Φ provides diindolymethane (DIM) and glucoraphanin as SGS. DIM promotes healthy estrogen metabolism and creates a better balance of estrogen metabolites (2-OH, 4-OH, 16-alpha-OH) through phase I cytochrome P450 enzyme induction and promotion of 2-hydroxylation.[19,20] The action of DIM is complemented by glucoraphanin, which supports long-term antioxidant activity and phase II detoxification of less-desirable estrogen metabolites and xenoestrogens.[21,22] Glucoraphanin and its metabolite sulforaphane are found to be effective, long-acting, indirect antioxidants and significant inducers of phase II detoxification enzymes.[23,24] These actions may help support healthy estrogen balance and may be crucial for the health of estrogen-sensitive tissue.*
Directions
as
References
- Gaby AR. Nutritional Medicine. Concord, NH: Fritz Perlberg Publishing; 2011.
- Colborn T, vom Saal FS, Soto AM. Developmental effects of endocrine-disruptingchemicals in wildlife and humans. Environ Health Perspect. 1993 Oct;101(5):378-84. Review. [PMID: 8080506]
- Alexander BJ, Ames BN, Baker SM, et al. Textbook of Functional Medicine. Gig Harbor, WA: The Institute for Functional Medicine; 2010.
- Singleton DW, Khan SA. Xenoestrogen exposure and mechanisms of endocrine disruption. Front Biosci. 2003 Jan 1;8:s110-8. Review. [PMID: 12456297]
- Nadal A, Ropero AB, Laribi O, et al. Nongenomic actions of estrogens and xenoestrogens by binding at a plasma membrane receptor unrelated to estrogen receptor alpha and estrogen receptor beta. Proc Natl Acad Sci U S A. 2000 Oct 10;97(21):11603-8. [PMID: 11027358]
- Liska DJ. The detoxification enzyme systems. Altern Med Rev. 1998 Jun;3(3):187-98. Review. [PMID: 9630736]
- Krinsky DL, LaValle JB, Hawkins EB, et al. Natural Therapeutics Pocket Guide. 2nd ed. Hudson, OH: Lexi-Comp; 2003.
- Pradhan SC, Girish C. Hepatoprotective herbal drug, silymarin from experimental pharmacology to clinical medicine. Indian J Med Res. 2006 Nov;124(5):491-504. [PMID: 17213517]
- Suzuki YJ, Aggarwal BB, Packer L. Alpha-lipoic acid is a potent inhibitor of NFkappa B activation in human T cells. Biochem Biophys Res Commun. 1992 Dec 30;189(3):1709-15. [PMID: 1482376]
- Baur A, Harrer T, Peukert M, et al. Alpha-lipoic acid is an effective inhibitor of human immuno-deficiency virus (HIV-1) replication. Klin Wochenschr. 1991 Oct 2;69(15):722-4. [PMID: 1724477]
- Bhattacharya A. Methylselenocysteine: a promising antiangiogenic agent for overcoming drug delivery barriers in solid malignancies for therapeutic synergy with anticancer drugs. Expert Opin Drug Deliv. 2011 Jun;8(6):749-63. [PMID: 21473705]
- Shilo S, Pardo M, Aharoni-Simon M, et al. Selenium supplementation increases liver MnSOD expression: molecular mechanism for hepato-protection. J Inorg Biochem. 2008 Jan;102(1):110-8. [PMID: 17804075]
- Brown MD. Green tea (Camellia sinensis) extract and its possible role in the prevention of cancer. Altern Med Rev. 1999 Oct;4(5):360-70. Review. [PMID: 10559550]
- Shankar S, Ganapathy S, Srivastava RK. Green tea polyphenols: biology and therapeutic implications in cancer. Front Biosci. 2007 Sep 1;12:4881-99. Review. [PMID: 17569617]
- Ferrer P, Asensi M, Segarra R, et al. Association between pterostilbene and quercetin inhibits metastatic activity of B16 melanoma. Neoplasia. 2005 Jan;7(1):37-47. [PMID: 15736313]
- Jurenka JS. Anti-inflammatory properties of curcumin, a major constituent of Curcuma longa: a review of preclinical and clinical research. Altern Med Rev. 2009 Jun;14(2):141-53. [PMID: 19594223]
- Choi H, Chun YS, Shin YJ, et al. Curcumin attenuates cytochrome P450 induction in response to 2,3,7,8-tetrachlorodibenzo-p-dioxin by ROS-dependently degrading AhR and ARNT. Cancer Sci. 2008 Dec;99(12):2518-24. [PMID: 19018768]
- Shoba G, et al. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998 May; 64 (4):353-6. [PMID: 9619120]
- Dalessandri KM, Firestone GL, Fitch MD, et al. Pilot study: effect of 3,3’-diindolylmethane supplements on urinary hormone metabolites in postmenopausal women with a history of early-stage breast cancer. Nutr Cancer. 2004;50(2):161-7. [PMID: 15623462]
- Kim EJ, Shin M, Park H, et al. Oral administration of 3,3’-diindolylmethane inhibits lung metastasis of 4T1 murine mammary carcinoma cells in BALB/c mice. J Nutr. 2009 Dec;139(12):2373- 9. [PMID: 19864400]
- Bolton JL, Thatcher GR. Potential mechanisms of estrogen quinine carcinogenesis. Chem Res Toxicol. 2008 Jan;21(1):93-101. Review. [PMID: 18052105]
- Keum YS. Regulation of the Keap1/Nrf2 system by chemopreventive sulforaphane: implications of posttranslational modifications. Ann N Y Acad Sci. 2011 Jul;1229:184-9. Review. [PMID: 21793854]
- Boddupalli S, Mein JR, Lakkanna S, et al. Induction of phase 2 antioxidant enzymes by broccoli sulforaphane: perspectives in maintaining the antioxidant activity of vitamins A, C, and E. Front Genet. 2012;3:7. Epub 2012 Jan 24. [PMID: 22303412]
- Sulforaphane glucosinolate. Monograph. Altern Med Rev. 2010 Dec;15(4):352-60. Review. [PMID: 21194251] p. 352.